
Who decides how long a second is? - John Kitching
Curated from: www.youtube.com
Ideas, facts & insights covering these topics:
5 ideas · 1.2K reads

The history of the second
The second as we know it was one introduced in the late 1500s when the Gregorian calendar began to spread across the globe alongside British colonialism.
The Gregorian calendar defined a day as a single revolution of the Earth about its axis. A day could be divided into 24 hours, each hour into 60 minutes, and each minute into 60 seconds.
Yet, the second was more a mathematical idea than a useful unit of time.

The growing importance of the second
The second only became important when society became interconnected through fast-moving railways where cities needed to agree on exact timekeeping.
By the 1950s, several global systems required every second to be accounted for with as much precision as possible.
Today, devices are synchronised to help us maintain a globally consistent time, from radio signal transmitters to satellites for global positioning systems.

Atomic clocks
As early as 1955, researchers started to develop atomic clocks, which relied on the laws of physics to establish a new foundation for timekeeping.
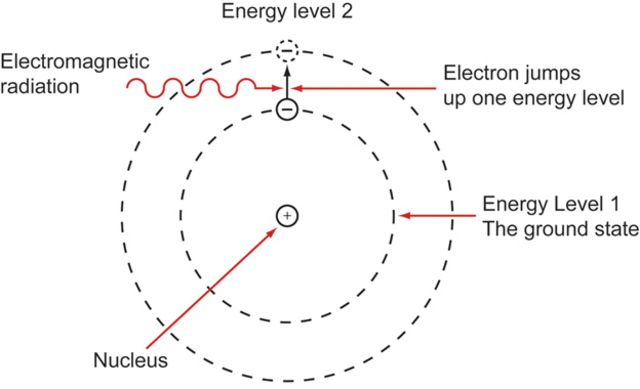
An atom consists of negatively charged electrons orbiting a positively charged nucleus at a consistent frequency. You can lightly disturb an electron's orientation if you expose an atom to an electromagnetic field such as light or radio waves.
If you briefly tweak an electron at the right frequency, the vibration will resemble a ticking pendulum and can tick for centuries.
Finding the right element
Before timekeeping could go fully atomic, countries had to decide which atom would work best.
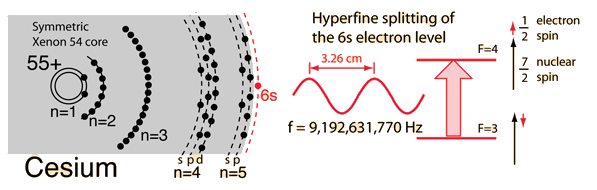
At the Thirteenth General Conference of the International Committee for Weights and Measures in 1967, researchers decided on Cesium-133.
The element conformed to the following specifications:
- It had a long-lived and high-frequency electron oscillation for precise, long-term timekeeping.
- It maintained a reliably measurable quantum spin
- It could easily vaporise.
The cesium standard
Determining how many ticks of a caesium atom were in a second, the conference used the most precise astronomical measurement of a second available at the time.
They started with the number of days in a year and divided it down. Compared to the atom’s ticking rate, one second was defined as exactly 9,192,631,770 ticks of a caesium-133 atom.


